
What was James Chadwicks atomic model called? John Dalton was the first to adapt Democritus’ theory into the first modern atomic model. The ancient atomic theory was proposed in the 5th century bc by the Greek philosophers Leucippus and Democritus and was revived in the 1st century bc by the Roman philosopher and poet Lucretius. According to atomism, matter consists of discrete particles. The word “atom” comes from the ancient Greek word atomos, which means indivisible. 
Each element has its own atomic number, which is equal to the number of protons in its nucleus.Ītomic theory originated as a philosophical concept in ancient India and Greece. Atoms themselves are composed of protons, neutrons, and electrons. What is the atomic model theory?Ĭhemistry is based on the modern atomic theory, which states that all matter is composed of atoms. This new idea dramatically changed the picture of the atom and accelerated discoveries in atomic physics. His findings were quickly accepted and Werner Heisenberg then showed that the neutron could not be a proton-electron pairing, but had to be its own unique particle - the third piece of the atom to be found. How did the discovery of the neutron change the atomic model? He is sometimes called the “father” of atomic theory, but judging from this photo on the right “grandfather” might be a better term. The idea that everything is made of atoms was pioneered by John Dalton (1766-1844) in a book he published in 1808. The protons behaved in exactly the manner they ought to if they had been hit by electrically neutral particles with a mass similar to the proton. 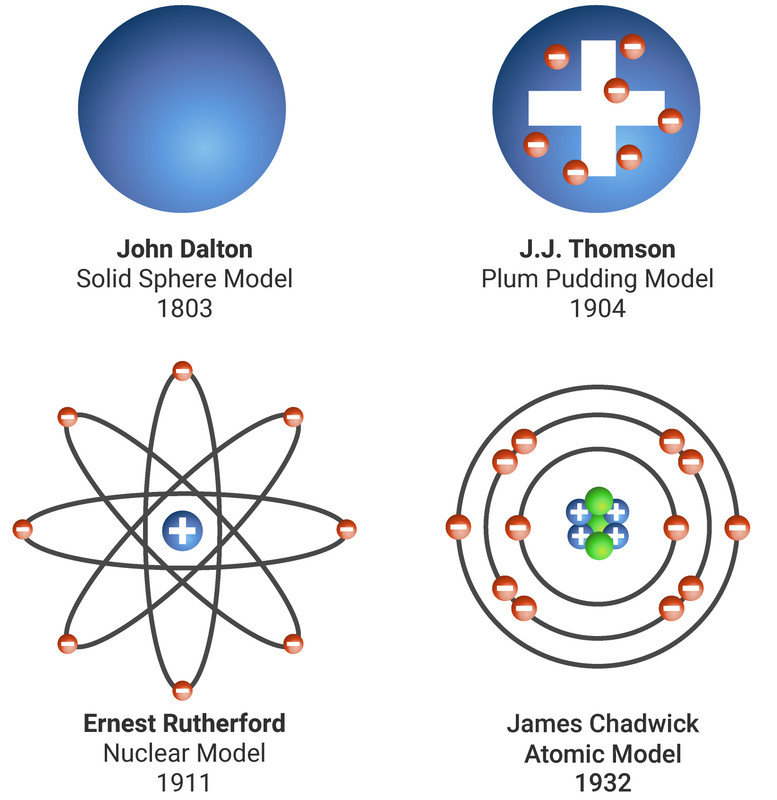
Protons were released by the wax and Chadwick made measurements of the protons’ behavior. 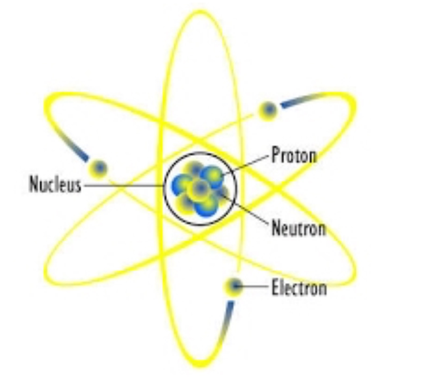
Using polonium as a source of (what he believed were) neutrons, he bombarded wax. How did James Chadwick find this new information? He was awarded the Hughes Medal of the Royal Society in 1932, and subsequently the Nobel Prize for Physics in 1935. In 1932, Chadwick proved the existence of neutrons, preparing the way towards the fission of Uranium 235 and the creation of the atomic bomb. Did James Chadwick help create the atomic bomb? For his mass measurement, Chadwick bombarded boron with alpha particles. Instead he measured everything else in the collision and used that information to calculate the mass. Therefore also coming to the conclusion that neutrons had a heavier mass than protons.To prove that the particle was indeed the neutron, Chadwick measured its mass. However it discharged protons, which meant that the particles had to be more massive than previously expected. This was when photons such as gamma rays, strike certain surfaces, they discharge electrons. Ĭhadwick could tell that the mysterious radiation was neutral because it was not deflected by electric or magnetic fields and it did not invoke the photoelectric effect, unlike standard gamma radiation. He was convinced that the beryllium was emitting neutrons. Chadwick, however did not believe that gamma rays could account for the protons from the wax. He then discovered that this radiation, upon striking Paraffin Wax (proton-rich surface), would dislodged some of the protons, coming to the conclusion it was gamma rays. In 1932, Chadwick carried out an experiment in which a sample of Beryllium was bombarded with alpha particles, which caused it to emit this mysterious radiation.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
